
Applied Science Letters


Background: Mesenchymal stem cells (MSCs) are a striking basis for intractable patients since they possess immunomodulatory/anti-inflammatory potential and multi-lineage differentiation capability. Several positive mediated effects have been indicated in MSCs based transplantation on preclinical and clinical application entailed for the treatment of many kinds of diseases. Main text: MSCs used in the treatment of various diseases comprise autoimmune disease, cancer, liver cirrhosis, cardiovascular diseases, and ischemic stroke. The paracrine factors of the exosomes involved in signal transduction by regulation and differentiation of new tissue during tissue injury repair. Robust progress in the isolation of multipotent progenitor cells, commonly defined as mesenchymal stromal/stem cells (MSCs), which have revealed an urge in cell therapy and regenerative medicine. Conclusion: Therefore the application of these MSCs derived exosomes obtained from several tissues has revealed to be beneficial for therapeutic application and publicized to be reliable for a cure in regenerative medicine. This review recapitulates the contemporary strategies implicated in the substantial role of MSC-derived exosomes as a remedy agent.
Role of Mesenchymal Stem Cells Derived Exosomes and Its Regenerative Potential
Padma Kanchi Ravi1*, Janardhana Papayya Balakrishna2, Lavina Dhawale2
1Department of Biotechnology, Sri Padmavati Mahila Visvavidyalayam, Padmvathi Nagar, Tirupati, Andhra Pradesh.
2Department of Biotechnology, Stellixir Biotech Private Ltd., Peenya 2nd Stage Industrial Area, Bangalore, Karnataka.
ABSTRACT
Background: Mesenchymal stem cells (MSCs) are a striking basis for intractable patients since they possess immunomodulatory/anti-inflammatory potential and multi-lineage differentiation capability. Several positive mediated effects have been indicated in MSCs based transplantation on preclinical and clinical application entailed for the treatment of many kinds of diseases. Main text: MSCs used in the treatment of various diseases comprise autoimmune disease, cancer, liver cirrhosis, cardiovascular diseases, and ischemic stroke. The paracrine factors of the exosomes involved in signal transduction by regulation and differentiation of new tissue during tissue injury repair. Robust progress in the isolation of multipotent progenitor cells, commonly defined as mesenchymal stromal/stem cells (MSCs), which have revealed an urge in cell therapy and regenerative medicine. Conclusion: Therefore the application of these MSCs derived exosomes obtained from several tissues has revealed to be beneficial for therapeutic application and publicized to be reliable for a cure in regenerative medicine. This review recapitulates the contemporary strategies implicated in the substantial role of MSC-derived exosomes as a remedy agent.
Keywords: MSCs, multi-lineage differentiation, exosomes, regenerative medicine, therapeutic application.
INTRODUCTION
Mesenchymal stem cells (MSCs) are the most substantially employed cell types in tissue repair as well as therapeutic potential in several human diseases [1]. MSCs are multipotent adult stem cells having immunomodulatory and regenerative properties [2, 3]; also they are characterized by minimal side effects and lack of rejection [4]. Since these somatic cells are regulated by paracrine factors which involve several proteins and peptides which forms intracellular signaling mechanism which revealed to possess an excellent immunomodulatory as well as multi-lineage properties [1]. “Roughly750 clinical trials were carried out employing MSCs which are listed at “www.clinicaltrials.gov” signifies its importance in the treatment of several clinical diseases. The Regenerative potential of the MSCs especially aims to reinstate the growth of injured tissues or organs. Organ transplantation is costly, there might be a lack of donors in such cases the suitable option was found to be MSCs, where there were fewer chances for immunological rejection [5].
The stem cells revealed to possess imprecise cell division potential as well as self-renewal ability and hence massively utilized in therapeutic medicine and stem cell therapy. The MSCs potential abilities included multi-lineage differentiation, migration to specific targets as well as immunomodulatory effects [6]. Chiefly, the stem/stromal cells are of two types: one is embryonic stem cells (ESCs) and the other one is Adult stem cells. Although it has been revealed in various study reports that the embryonic stem cells (ESCs) are pluripotent cells which have been disengaged from the inner cell mass of blastocysts, on the other hand, the adult stem cells persisted to subsist in adult tissues. MSCs displayed their paracrine signaling effects through growth factors surrounding it. Several growth factors involved around the MSCs included the vascular endothelial growth factor (VEGF), hepatocyte growth factor (HGF), angiopoietin, and epidermal growth factor (EGF) and nitric oxide, which initiated for cellular differentiation [7]. Hence, these paracrine effects enabled the growth factors to distinguish into diverse types of cells including, ligaments, bone, tendons, cartilage, muscles, and later to neurons.
Mesenchymal stromal cells materialized as an imminent solution for any sort of tissue injury and healing of wounds. In our review, we will portray current studies of MSCs derived exosomes and their importance in cancer development, transplantation as well as the mechanism that MSC-exosomes transport cancer drug resistance. However, we also have communicated about their application in clinical diagnostics as well as treatment.
Stem Cell Exosomes and its characteristics
Stem cells are unspecialized cells yet, have remarkable self-renewal capability to maintain homeostasis as well as tissue repair. Since they have a loaded cargo of proteins such as ˃160 miRNA and ˃900 unique proteins resulting in the modification of many activities of target cells through diverse pathways [8]. The proteins involved in mesenchymal stromal cells modulate in tissue repair and exhibits therapeutic functions in a paracrine manner rather than a cellular manner. MSCs are the principal model for the accrual production of exosomes for drug delivery [9]. Since it has low immunogenicity, MSCs can repress the events of several immune effector cell types and enhance immune regulatory functions as depicted in (Figure-1). Relatively smaller size exosomes are transported through the blood flow to distant organs for interaction with cells. The potentiality of exosomes depends upon the components within them. It has been exposed that exosomal proteins, predominantly are the amalgamation of endosomes, plasma membrane, and cytosol proteins, along with few other intracellular protein organelles [10].
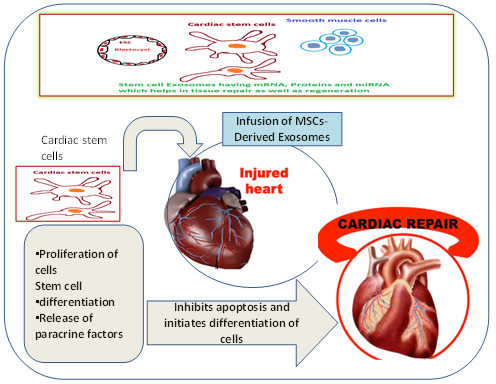
Figure 1: Characteristics of Mesenchymal stem cells Involved in Tissue repair
Effects of MSC-EVs
Following current studies, stem cell transplantation chiefly revealed a positive response in the healing of wounds by a paracrine mechanism. Nevertheless, the exosomes are the major components secreted through this mechanism which contributed to the recovery of wounds [11]. Shabbir et al. exhibited that human umbilical vein endothelial cells (HUVECs) displayed the ability to internalize MSC-DEs. However, they stated that enhancing the concentration of MSC-DEs provoked extensive vascular tube formation. This in turn persuaded the MSC-DEs to carry active STAT3 transcription factor, which is indulged in the transcription of VEGF, HGF, and IL-6. In addendum, MSC-DEs enhance the growth and migration of normal as well as diabetic wound fibroblasts. Zhang et al. also revealed that exosomes from iPSC-MSCs provoked the regeneration of newly shaped vessels and resulted in their maturation in wound sites [12][Shown in figure-2].
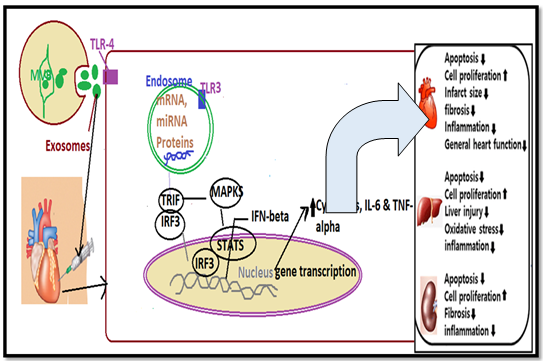

Figure 2: MSC-DEs binds to toll-like receptors and urge tissue regeneration
Role of MSC-exosomes in angiogenesis
Several studies have been displayed on the role of MSC-exosomes in angiogenesis, it has been demonstrated that the cancer cells derived from the exosomes possess interleukin-6 (IL-6) as well as sturdy pro-angiogenic factors, vascular endothelial growth factor (VEGF) and several other molecules which enables to enhance organization and endothelial cell in tubule-like structures [13]. Diverse studies have illustrated that MSCs play an imperative role in angiogenesis, even though the function of MSC-exosomes in angiogenesis is displayed to be controversial but various other studies have proposed that the external secretion of the body has been produced by blood vessels. Even though there are also several data presenting MSC-exosomes secretion which apprehends the formation of VEGF. However, the increased concentration of MSC-exosomes effectually reduced tumor growth and angiogenesis in the beginning and posed that even the anti-tumor effects of MSC-exosomes were not scrawny eventually. Hence, MSC-derived exosomes posed to be a valuable anti-angiogenetic agent for anti-tumor treatment as shown in (Figure-3).
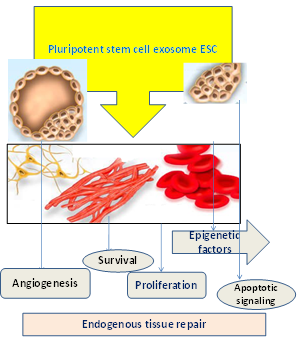
Figure 3: Endogenous tissue repair
Characteristics and Purification of Exosomes
Following protein analyses, there are an estimated 1600 proteins in fluid-derived exosomes. Mostly exosomal proteins comprise heat-shock proteins (HSP70, HSP60, and HSP90), tetraspanins (CD9, CD63, and CD81), and tumor susceptibility gene 101 (TSG101), and also have several proteins as diagnostic markers during pathological conditions. An alternative composition of exosomes is RNA. Exosomes showed to change the fate of target recipient cells by transporting mRNAs and miRNAs. They also provided as diagnostic biomarkers in specific diseases as well as in cancers. Lipid composition analyses have been demonstrated that exosomes are enriched with sphingomyelin, cholesterol, phosphatidylserine, and ceramide. Table-1 [14] reveals the characteristics of exosomes which included the definition, markers, size, isolation methods, identification, as well as storage conditions.
Table-1: Characteristics of Exosomes
|
Markers |
Tetraspanins, Alix, CD9, CD81, CD63, Hsp60, Hsp70, Hsp90, Tsg101, lipid rafts, Rab5b flotillin |
|
Size |
Ultracentrifugation, HPLC, ultrafiltration, sequential filtration, SEC, F4, mass spectrometry, precipitation methods, microfluidic |
|
Isolation methods |
Atomic force microscopy, scanning electron microscopy, nanoparticle tracking analysis, flow cytometric analysis, transmission electron microscopy, western blotting, ELISA |
|
Storage condition |
-20°C (without affecting the size of exosomes) |
Currently, MSCs are recognized as the sole human cell type to have the capability to increase exosomes production. During the condition of myocardial ischemia or reoxygenation injury, the utilization of MSC-derived exosomes was first demonstrated in animal models specifically mouse in 2010. The characterization of exosomes has also been initiated to associate with its cell origin. For instance, almost all the MSC-derived exosomes have expressed markers such as CD63 CD9 and CD81 [15]. Nevertheless recognizing the source of exosomes and isolating them particularly from the extracellular matrix is an exclusive way of current approaches. Later purification of those isolated exosomes by ultracentrifugation at 100,000×g is the most broadly utilized method.
Clinical application of MSCs isolated from different sources
Mesenchymal Stem cells can be secluded from several sources in the human body, the choice of which should be preferably based on their practical, logistical as well as In-vitro characteristics. Presently, the abundant sources of MSCs are the bone marrow and adipose tissue (AT). Nevertheless, the MSCs can notionally be attained from almost any tissue present within the human body, even though there are several practical limitations relating to the obscurity, invasiveness of the procurement process, and several donor characteristics. To obtain BM-MSCs, for instance, it might result in pain, bleeding, or infection. Hence harvesting the MSCs from BM source represents to be more problematic than harvesting cells from peripheral blood or surgical remnants such as AT or birth-derived tissues [16]. Table-2 describes existing sources of MSCs along with their characteristics features, their importance as well as use, and its limitation in relationship with clinical applications.
Table 2: Comparison of applications of Mesenchymal stem cell sources with MSCs derived Exosomes
|
Source |
Method of procurement |
Advantages |
Disadvantages |
Clinical applications |
References |
|
Adipocytes Astrocytes Cardiomyocytes Chondrocytes Hepatocytes Mesangial cells Muscle cells Neurons Osteoblasts Stromal cells Embryonic tissue |
Isolated from Bone Marrow aspirate |
Stem cells isolated from these sources have the potential to differentiate into Adipose tissue-MSCs.
Multiple clinical trials have confirmed the safety and effectiveness of this type of stem cell.
|
The procurement of these cells from their source is often painful and carries the risk of infection. Cell yield and differentiation potential is dependent on donor characteristics i.e. age |
Generation of pancreatic cellsIn-Vitro Treatment of Orthopaedic conditions characterized by large bone defects, including articular cartilage repair, osteoarthritis, and rheumatoid arthritis Bone Marrow-MSCs promote osteogenesis. |
[17] |
|
Adipocytes Chondrocytes Osteocytes Muscle cells
|
Isolated by Liposuction, lipoplasty or lipectomy |
A large No. of stem cells can be isolated from adipose tissue rather than in comparison with BM. |
But have less Osteogenic and Chondrogenic potential in comparison with BM-MSCs |
Cosmetic and Dermatological application Successfully used in the treatment of skeletal muscle injuries. Potential in cell-based therapy |
[18] |
|
Rat BM-MSCs cultured in Exosome free serum |
Isolated EV by UC/100μg total exosome protein |
MSCs cultured in spheroid showed higher efficacy and safety profiles, and decreased the expression of integrins, resulting in increased secretion of EVs |
Characteristics of EVs, as well as phenotypes of stem cells, could be affected by mechanical forces |
↑Angiogenesis and Neurogenesis Immunomodulation |
[19] |
|
Human AT-MSCs cultured in Serum-free media with brain extracts |
Isolated EV by UC/0.2mg/kg/dose per animal |
Cultivation of MSCs with either serum obtained from stroke patients, or treatment of ischemic brain extracts on culture media, could activate restorative properties of MSCs |
low-yield by conventional cell culture systems. |
↑Angiogenesis and Neurogenesis Immunomodulation |
[20] |
Cargos of Extracellular vesicles
Commonly, EVs have been described as small membrane vesicles and comprising of exosomes, microvesicles, and apoptotic bodies. The Extracellular vesicles are broadly established based on their basis of biogenesis. Under the EVs, classes include Exosomes and microvesicles. Nevertheless, these vesicles are surrounded by a phospholipid membrane and contain various specific types of cell components, such as proteins, lipids, RNA, and metabolites. Previously there has been little confusion existed in the literature respect to the terms “exosomes” and “microvesicles”. Conversely, the most comparatively and broadly studied EVs are none other than Exosomes, characterized basically of 50–150 nm-sized derivatives. Commonly, they result in by formation of internal budding of the endocytic membrane, designated as multivesicular bodies (MVBs) and these exosomes are consequently secreted to the extracellular environment as membranous vesicles nature and relatively blends with the plasmalemma [21].On comparison, the microvesicles have been produced directly through budding of the plasmalemma and vary in the range of 100–1000 nm in diameter. Even though the MV is released, it does not involve exocytosis. However, these Microvesicles have commonly been referred to as microparticles. The EVs have a key role in the transfer as well as transportation of bioactive molecules, such as proteins and RNA, in-between cells. Additionally, Although EVs released inside the cell later can change their compositions inside in response to cell activation, for instance as hypoxia, irradiation, injury, and cellular stress. Hence, EVs have displayed to engage in revealing their significant roles for a broad range of pathological conditions, including lung diseases, by acting as cargo.
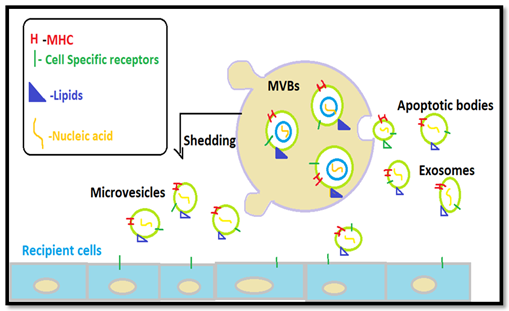
Figure 4: EVs origin and Release
Synthesis of EV
EVs are a general term that characteristically referred to as the heterogeneous vesicles which are specifically released from the secretory cells. Nevertheless, the secretory EVs contained predominantly of cellular proteins, DNAs, RNAs of cells and hence regarded by various names such as exosomes (30–200 nm) and usually also termed as microvesicles (200–1000 nm) and generally named as apoptosomes (1– 10µm) depending on their size [22]. Amongst those, exosomes and microvesicles are especially released from biological cells that are perceived to involve in many processes, such as proliferation, differentiation, and angiogenesis, and are known to act as a means of signal transduction. These Exosomes and microvesicles regularly arise from the plasma membrane. Once the recruitment of cytoplasmic proteins and nucleic acids results thereby the endosomal sorting complex required for transport (ESCRT) which is generally mediated by ADP ribosylation factor 6 [ARF6] and phospholipase D2 [PLD2]. Lipid flipping happens, and as a result budding of the membrane takes place.
MSCs and its clinical application
The advantageous therapeutic effects of MSCs were shown in several studies and relatively came into action after the primary clinical trial which was exhibited in 1995. Currently, the data from literature reports of clinical trials have revealed numerous clinical applications of MSCs for a wide variety of diseases such as involving of acute myocardial ischemia, lung fibrosis and in case of spinal cord injury and several neurogenetic disorders; like Alzheimer’s disease as well as stroke and amyotrophic lateral sclerosis [23]. Although previous studies on hepatic regeneration have been demonstrated wherein cases due to carbon tetrachloride the liver was injured. Therefore with the utilization of human placenta-derived MSCs significantly retrieved tissue regeneration.
Presently, the application of MSC-derived exosomes has been recommended as an inimitable strategy in regenerative medicine and tissue engineering since there is no immune rejection and tumor formation based on MSC properties [24]. Moreover, the make use of exosomes is suitable in stipulations of fabricating as undifferentiated MSCs, displaying to have a long lifespan compared to that of other types of cells. Commonly, MSC-derived exosomes carry the complex biologically active molecules including proteins and mRNAs. However, these exosomal contents are combined with several cellular and biochemical processes and help in cell-to-cell communication, tissue regeneration, metabolism as well as immune modulation as depicted in (Figure 5). Therefore, MSC-derived exosomes have been considered as a consensus tool to perceive various molecules produced from various cellular responses via interaction with diverse cell types and employed as a chief therapeutic agent.
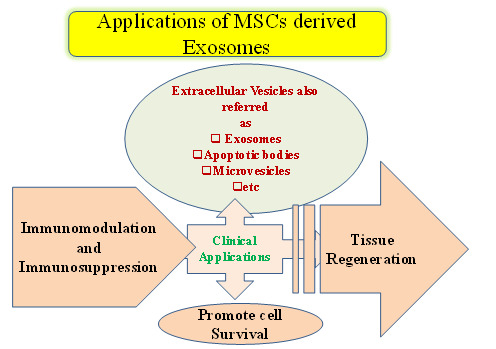
Figure 5. Applications and actions of mesenchymal stem cells (MSCs).
Future Prospective
Taking into account the previous studies, we have focussed on the current development and latest knowledge of MSC-derived EV-based therapeutics. MSCs have been the best as well as interesting because of their therapeutic effects which resulted in immune modulation and tissue remodeling. Even though, the mechanisms by which MSCs encourage modest inflammation and injury that are not reasonably understood and might involve distinct multiple pathways which revealed to be mediated and released by several soluble factors as well as EVs. MSC-derived EVs illustrated many possible advantages in comparison with cell-based therapies and hence showed much of their application in regenerative medicine. Several growing bodies of evidence indicated that MSC-derived EV-based therapies for inflammatory lung diseases, Cardiac disease are evolving to become viable treatment options. Virtually earlier studies indicated that MSC-derived EVs have displayed to present their effects via the horizontal transfer of proteins, mRNAs, and miRNAs.
Since the MSCs-derived exosomes are small in size, simple to modify and very less toxicity are the basic advantages of exosomes in comparison with cell therapy. Hence, exosomes are utilized as an appropriate choice for whole-cell therapy and can even be employed as carriers of therapeutic agents, both in medical regenerative medicine as well as in cancer therapy. Earlier to entering through clinical trials, several concerns about stability, surface changes for targeting, and multiple effects on signaling pathways as well as effective compounds that were used in pre-clinical examinations have to resolve.
CONCLUSION
Several studies reported the advantages of MSCs derived exosomes. Therefore, mass fabrication of exosomes made possible by good manufacturing practice grade for safe and efficient clinical applications. However, effectual lead delivery of exosomes into targeted cells or tissues is crucial for promising therapeutic efficacy. The comprehension of cell-to-cell communication via microvesicles including exosomes is also significant to maximize the effects of exosome-mediated approaches in the field of stem cell therapy and also in regenerative medicine. The developing technology and observation through clinical testing using MSC-based exosomes have led one step closer to the more rapid development of stem cell-based therapies. The conclusion of our present review study in comparison with several other study reports stresses the therapeutic potential of MSC-derived exosomes which seems to present promising new stem cell-based cell-free agents for many intractable and rare diseases.
Abbreviations
ARF6: ADP ribosylation factor 6
PLD2: phospholipase D2
AT: Adipose tissue
BM-MSCs: Bone marrow- Mesenchymal stem cells
EGF: Epidermal growth factor
ELISA: Enzyme-Linked Immunosorbent Assay
ESCs: Embryonic stem cells
ESCRT: Endosomal sorting complex required for transport
EV: Extracellular Vesicles
HGF: hepatocyte growth factor
HUVECs: human umbilical vein endothelial cells
MSCs: Mesenchymal stem cells
MSC-DEs: Mesenchymal stem cells-Derived Exosomes
MVBs: Multivesicular bodies
MSC-EVs: Mesenchymal stem cells-Extracellular Vesicles
PSC-MSCs: Pluripotent stem cells- Mesenchymal stem cells
VEGF: Vascular endothelial growth factor.
REFERENCES
